Abstract
Many clinical and animal studies have shown that certain dietary components exert anti-inflammatory properties that aid in the amelioration of chronic inflammatory diseases. Among the various proposed channels through which dietary components affect immune responses, regulatory T-cells (Tregs) are emerging as key targets for the dietary prevention of chronic inflammatory diseases. In this review, immunoregulation by Tregs is briefly described, followed by a summary of recent advances and possible applications of techniques for the study of Tregs. In addition, this review provides an overview of the current knowledge on Treg regulation by certain dietary components, including vitamins, omega-3 polyunsaturated fatty acids, and polyphenols. The caveats of previous studies are also discussed in order to highlight the distinctions between dietary studies and immunological approaches. Consequently, this review may help to clarify the means by which nutritional components influence Tregs.
Keywords: nutrition, immunology, regulatory T-cells, Tregs, anti-inflammatory
4. Dietary Regulation of Tregs
4.1. Vitamins
Following dietary consumption and absorption in the intestines, water-insoluble vitamin A (all-trans-retinol) is carried by cellular retinol binding protein (CRBP) in an aqueous environment to be transported into the cytoplasm. Subsequently, retinol is oxidized to retinal by retinol dehydrogenase, and then retinal is further oxidized to retinoic acid by retinal dehydrogenase [25]. With respect to the role of vitamin A in the development of Tregs, Bai et al. [26] demonstrated that the population of Tregs derived from biopsies of ulcerative colitis patients increases following ex vivo culture in the presence of retinoic acid, a potent metabolite of vitamin A. In the same study, using a chemical (2,4,6-trinitrobenzene sulfonic acid, TNBS)-induced murine colitis model, it was further elucidated that dietary vitamin A ameliorated colitis, which is accompanied by an increased population of Tregs. Wu et al. [27] further reported that intraperitoneal administration of all-trans retinoic acid aided in the attenuation of airway inflammation by inducing Treg development in a model of experimental allergic asthma. Moreover, a dietary study demonstrated that retinal intervention in mice upregulated Tregs, which further assisted in the treatment of autoimmune inflammatory disorders, including rheumatoid arthritis [28]. Overall, these animal studies indicate that vitamin A and its metabolites affect populations of Tregs, thereby suppressing chronic inflammatory diseases. However, it remains unclear whether vitamin A affects Tregs directly, indirectly, or both. Indeed, Chang et al. demonstrated that dendritic cells also promote the generation of Tregs in response to retinoic acid, at least in vitro [29], exemplifying that in vivo models are more complicated for elucidating the mechanism of action of dietary components.
With respect to water-soluble vitamins, Kunisawa et al. [30] showed that Tregs express high levels of vitamin B9 (folic acid) receptor on their cell surfaces. Furthermore, vitamin B9 was demonstrated to be a survival factor for Tregs; in a vitamin B9-deficient culture, naïve CD4+ T-cells successfully differentiated into Foxp3+ Tregs but failed to survive. Moreover, it was found that mice fed a vitamin B9-deficient diet exhibited a decreased number of Tregs in the small intestine, where vitamin B9 is absorbed.
4.2. Dietary Fatty Acids
A significant quantity of data has indicated that dietary omega-3 polyunsaturated fatty acids (PUFA) may prevent or ameliorate chronic inflammatory diseases, including inflammatory bowel diseases [31,32,33,34]. These studies have identified multiple anti-inflammatory mechanisms of omega-3 PUFA: cytokine production, antagonism to omega-6 PUFA metabolism, binding to nuclear receptors as ligands, the alteration of signaling protein acylation, and the modulation of signaling platform lipid rafts in various immune cell models. With respect to CD4+ T-cell mediated inflammatory responses, several studies using dietary intervention with either purified omega-3 PUFA or fish oil (which is rich in omega-3 PUFA) demonstrated that CD4+ T-cell functions, as assessed by cytokine production and proliferation, were suppressed in both humans [35,36,37] and experimental animals [38,39].
As for the effect of omega-3 PUFA on Tregs, it was shown that the omega-3 PUFA abundant in fermented fish oil enhanced the development of Foxp3+ Tregs in vivo [40]. Moreover, intraperitoneal injection of eicosapentaenoic acid (EPA), an omega-3 PUFA, resulted in prolongation of graft survival in a murine transplant model, accompanied by an increased population of Tregs [41]. However, those studies do not conclusively demonstrate a direct function of omega-3 PUFA on the differentiation and/or function of Tregs, given the caveat that in vivo administration of omega-3 PUFA can affect diverse types of accessory cells. Indeed, it was found that dendritic cells cultured in docosahexaenoic acid (DHA, an omega-3 PUFA)-rich conditions facilitated the development of Tregs, at least in vitro [42], indicating an indirect modulation of Treg development by omega-3 PUFA. However, despite the increase in the development of Tregs, the favorable effect of omega-3 PUFA on Tregs is still controversial because it has also been observed that DHA inhibited the suppressive effect of Tregs on effector T-cell proliferations in a dose-dependent manner in vitro [43].
4.3. Dietary Polyphenols
Dietary polyphenols are well known for their antioxidant properties, which can further suppress inflammatory responses by reducing nitric oxide. In addition to their archetypal antioxidant capacities, immunoregulatory effects of select polyphenols on Tregs have been characterized as follows. Wang et al. [44] reported that ex vivo development of CD4+ Foxp3+ Tregs was induced in the presence of the flavonoid naringenin in part via activation of the aryl hydrocarbon receptor, a transcription factor. As well, increased Treg differentiation suppressed the proliferation of effector T-cells. In addition, dietary naringenin was found to suppress cholesterol-induced systemic inflammation, metabolic dysregulation, atherosclerosis, and allergen-induced airway inflammation [45,46], although it was not clear whether those inhibitory effects were Treg-dependent. In a mouse model of T-cell mediated inflammatory disease of the central nervous system, dietary intake of epigallocatechin gallate (EGCG), a catechin derivative, exerted a favorable effect, in part by increasing the Treg population in the spinal cord [47]. Furthermore, fermented grape marc (FGM) was found to promote Treg differentiation of human CD4+ T-cells [48].Go to:
5. Conclusions
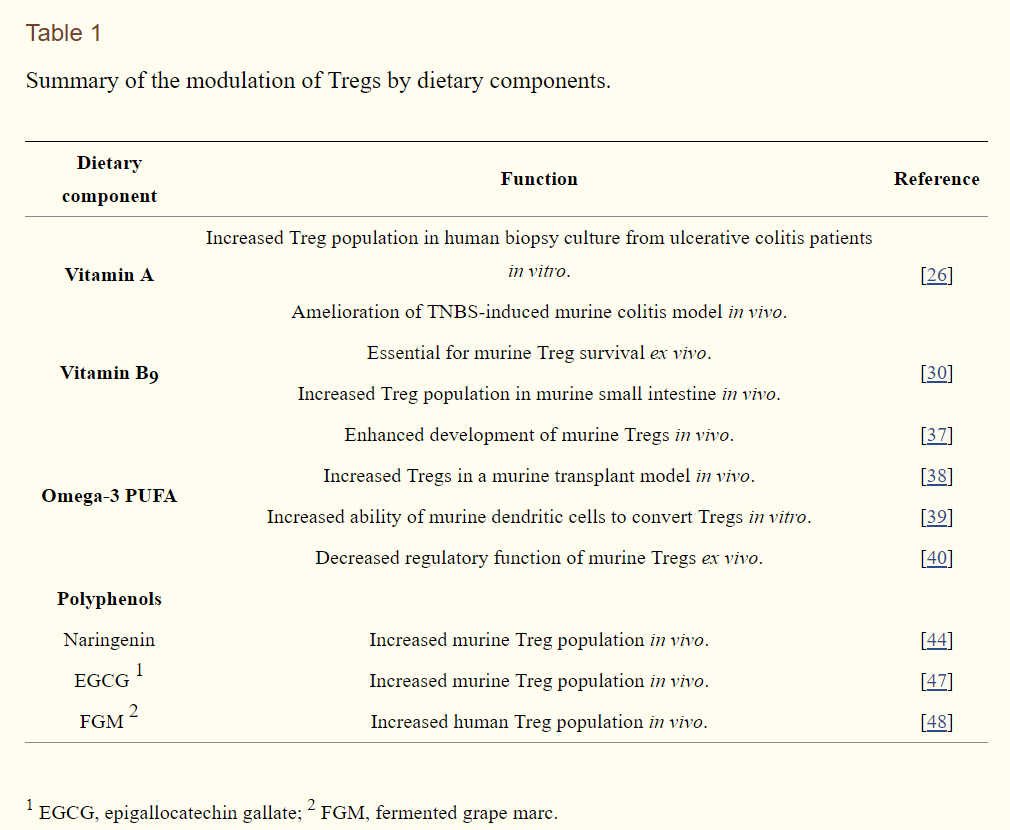
Due to the relatively short history of studies on Tregs, investigations of the direct effects of a variety of dietary components on Treg conversion are limited at present (summarized in Table 1 and previously reviewed elsewhere [49]). It seems that a majority of the anti-inflammatory functional studies have been conducted in vivo by observing physiological outcomes, such as symptoms of chronic inflammation and accumulation of certain immune cell types. Even though in vivo studies are indispensable for nutritional studies, simpler model systems must be developed to dissect the detailed mechanisms by which dietary components regulate physiological systems. Therefore, further studies using more sophisticated and appropriate in vivo model systems are needed to draw solid conclusions. In this regard, as introduced in this review, the protocol for Treg differentiation from naïve CD4+ T cells could be effectively utilized to determine the direct effects of a variety of promising dietary Treg modulators present in foods.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3847731/
Published: October 28th 2013

One Reply to “Advances in Nutritional Research on Regulatory T-Cells”