




Reflections





Are you among the thousands of people who are struggling with Lyme disease? If you are, you will understand just how painful and debilitating it can be.
According to the Center For Disease Control, there are around 30,000 new Lyme disease cases reported in the U.S each year. However, this is only a small proportion of the 300,000 or so people who are thought to be diagnosed each year. 1
In the UK, the disease is also not unheard of, with around 3,000 cases reported annually.
In Australia, The Lyme Disease Association of Australia estimates that around 300,000 Australians live with the disease.
Since the Australian government refuses to accept that Lyme disease can be contracted in the country, only two weeks of treatment are provided under the Medicare system. This means that many Lyme patients are forced to seek medical treatment overseas at specialist centers such as the Budwig Center here in Spain.
Lyme disease is a tick-borne infectious disease caused by the spirochete bacterium Borrelia Burgdorferi, which is usually transmitted to humans through a tick bite. Mosquitoes, spiders, and mites may also transmit it.
Lyme disease is not contagious, and a person can only be infected if bitten by an infected tick. It is also important to note that ticks do not generally transmit the disease until they have been attached to the host for at least 36 hours.

The most common places that tick bites occur are in forests, woodland, or health areas where you find tick-carrying animals such as deer or mice.
Typically, an incubation period can last from between two days and three and a half months. The bacteria does not always cause disease immediately. It is thought that bacteria can lie dormant and, although present, will not cause any symptoms. However, bacteria still has the potential to cause disease at a later stage.
Those with Lyme disease can exhibit similar symptoms to those with Fibromyalgia and Chronic Fatigue Syndrome in its latter stages.
Although there are thought to be around 100 different Lyme disease symptoms, these can vary widely from person to person. The most common are:-

In around 70% of cases, patients experience a rash on the skin in the shape of a “bulls-eye.” A large red ring on the outside, with a red circle in the middle. The rash is commonly referred to as Erythema Migrans or an EM Rash.
If left untreated, the infection can spread to the joints, the heart and may even disrupt the central nervous system, peripheral nervous system, and cardiovascular system. In some extreme cases, Lyme disease can result in Pericarditis (Inflammation of external heart tissues) or death from heart blockage.
There are two main tests for diagnosing Lyme disease:
If both of these tests return as positive, this is usually a clear sign that Lyme disease is the cause. Unfortunately, Lyme testing is not 100% accurate, with many doctors rule out the disease altogether, sometimes without any testing.
At the Budwig Center, we use the Vega test, which is a much better option and has a 93% success rate in identifying Lyme disease.
Judging by what we have heard from our own patients and reports on the internet, a vast number of physicians have somewhat limited knowledge when it comes to diagnosing Lyme disease. We often hear patients being told that the symptoms are related to other conditions such as ME/CFS or Fibromyalgia.
This is probably why the disease is also known as the “great imitator,”â as the symptoms are similar to so many other chronic conditions.
There are some tips which can help you to decrease the possibility of catching a tick bite. It is recommended to wear long pants and long-sleeved shirts when you are in forests, woodland, or health areas where you find tick-carrying animals such as deer or mice and use insect repellent. It is also vital that you remain vigilant and check yourself, your children, and your pets for ticks. Some assume that you can’t get infected if you’ve had Lyme disease before; however, this is not the case. If you do notice a tick, remove it with tweezers. It is important to check closely whether all tick parts have been removed.
Those treated with antibiotics in the early stages of a Lyme infection usually recover completely and in a relatively short space of time.
Unfortunately, the same cannot be said for around 20% of patients who will continue to experience persistent and recurring symptoms. These patients are classed as having PTLDS or Post Treatment Lyme Disease Syndrome.
Post-Lyme disease syndrome symptoms are similar to the symptoms that occur during the early stages. These symptoms include difficulty sleeping, fatigue, joint and muscle aches, short-term memory issues or difficulty concentrating, pain, and swelling in the larger joints, such as elbows, knees, shoulders, or speech issues. It’s fair to say that Lyme disease is one of the most challenging conditions to recover from fully, especially when at a more advanced stage.
Here at the Budwig Center clinic, our experienced therapists have successfully treated many Lyme disease patients over the last 10 years using BioMagnetism or BIO-MAGNETIC THERAPY.
Those treated with antibiotics in the early stages of a Lyme infection usually recover completely and in a relatively short space of time.
Unfortunately, the same cannot be said for around 20% of patients who will continue to experience persistent and recurring symptoms. These patients are classed as having PTLDS or Post Treatment Lyme Disease Syndrome.
Post-Lyme disease syndrome symptoms are similar to the symptoms that occur during the early stages. These symptoms include difficulty sleeping, fatigue, joint and muscle aches, short-term memory issues or difficulty concentrating, pain, and swelling in the larger joints, such as elbows, knees, shoulders, or speech issues. It’s fair to say that Lyme disease is one of the most challenging conditions to recover from fully, especially when at a more advanced stage.
Here at the Budwig Center clinic, our experienced therapists have successfully treated many Lyme disease patients over the last 10 years using BioMagnetism or BIO-MAGNETIC THERAPY.
UK based practitioner : https://thesouthdownclinic.co.uk/home/therapies/biomagnetic-pair-therapy/
Chooselife : My dear late friends Don and Carol Croft were the inventors and pioneers of Orgonite, one of their profound inventions was the Orgonite Zapper (Hulda Clark based technology, with Orgonite block added):
https://duckduckgo.com/?q=terminator+zapper+lyme+disease&ia=web
(no idea if the above Zapper site is better/worse than an Orgonite based Zapper, as it has been too many years since I mixed in these circles, this website has no affiliation with these or any other sellers, Don and Carols website was www.worldwithoutparasites.com )
Throw off your mental chains, choose life!
Wenju Lu, Xiaoqing Liu, Tao Wang, Fei Liu, Airu Zhu, and Yongping Lin
Patients with coronavirus disease 2019 (COVID-19) exhibit a spectrum of respiratory symptoms like cough and dyspnea.1–3 Airway mucus is an adhesive viscoelastic gel composed mostly of high-molecular-weight mucous glycoproteins and water, which is important in maintaining lung function and health, pathological mucus hypersecretion may cause airway obstruction and lead to respiratory distress. Mucin (MUC) glycoproteins are the major macromolecular components of mucus, which are classified into two major types: the gel-forming secreted MUC5AC and the membrane-tethered MUC1.4 Here, with an attempt to understand the lung changes, we sought to provide a delineation of the components of airway mucus from patients with COVID-19.
To clean airway obstruction, respiratory tract mucus was aspirated and collected via bronchoscopy from COVID-19 patients with a critical illness, and optical coherence tomography (OCT) was applied via bronchoscopy to obtain cross-sectional images of the bronchiole. For healthy control, sputum was induced by inhaled hypertonic (3%) saline solution delivered with an ultrasonic nebulizer. After collection, sputum was processed as previously described for components analysis.5 Medical history, and clinical and laboratory data of the participants were extracted from electronic medical records. The study was approved by the Ethics Committee of the First Affiliated Hospital of Guangzhou Medical University (No. 2020-65). The requirement for informed consent was waived because the study was observational and the family members were in quarantine.
MUC5AC (sc-21701; Santa Cruz, Shanghai, China) and MUC1 (sc-6827; Santa Cruz) in airway mucus were measured using enzyme-linked immunosorbent assay, and MUC1-cytoplasmic tail (CT, clone EP1024Y; Abcam, Shanghai) levels were measured by Western blot analysis as described previously.5 The levels of MUC5AC and MUC1 were normalized to their average signal reading of a healthy control group.
A total of 16 patients with COVID-19 were included in this study, the clinical characteristics of the recruited subjects were shown in Table S1. There was no significant deviation in the distribution of age, or sex between the cohorts of case and control subjects. All the patients with COVID-19 were admitted to the intensive care unit because of low oxygenation index (199 ± 23 mm Hg), and 79% of them received mechanical ventilation. Blood laboratory tests showed elevated inflammatory indexes including leukocyte count, C-reactive protein, and interleukin-6 in most of the patients with COVID-19 (Table S2).
OCT indicated clear bronchiole in healthy controls (Figure 1A) and mucus retention in the bronchiole of patients with COVID-19 (Figure 1B). A volume of 1-8 mL white to gray sputum with high viscosity (Figure 1C) was aspirated from the respiratory tract of 16 patients with COVID-19. Induced sputum from healthy control was clear and transparent with low viscosity. Compared to healthy control, airway mucus from patients with COVID-19 had a higher level of MUC5AC (Figure 1D), MUC1 (Figure 1E), and MUC1-CT fragment (Figure 1F). However, there were no significant differences in the concentration of total protein, sodium, or chloride in the airway mucus from patients with COVID-19 when compared to healthy control (Table S3).

Although more than half of patients with COVID-19 presented with a dry cough,6 this study provided direct evidence showing mucus retention in the small airway of patients with COVID-19, and patients were not able to expectorate by themselves and need bronchoscopy aspiration to help them to clean respiratory tract. The sputum from these patients with COVID-19 was viscous, which is not surprising as MUC5AC levels are extremely high, hyperconcentration of this gel-forming MUC dehydrates airway surfaces and causes mucus adhesion, which may contribute to airflow obstruction and respiratory distress. Clearance of airway mucus is an important way to increase oxygen and carbon dioxide exchange, bronchoscopy aspiration of airway mucus was used in all our patients to relieve hypoxia. In our center, all the 16 critical ill COVID-19 patients recovered and were discharged from hospitalization, which may attribute to our aggressive clearance of the respiratory tract. It is of note that bedside bronchoscopy may not be available in some hospitals as the medical resources are limited during the COVID-19 pandemic, carbocisteine has been reported to inhibit airway MUC5AC secretion, which could be used to reduce sputum viscosity and elasticity in patients with COVID-19. In addition, hydration of sputum by aerosolized hypertonic saline solutions or mannitol, and dilation of bronchi via aerosolized salbutamol may facilitate sputum expectoration.
MUC1 is a membrane-tethered MUC expressed on the apical surface of epithelial cells.7, 8 Since MUC1-CT fragment is on the cytoplasmic side of the cells, the elevated sputum CT fragment in patients with COVID-19 could come from detached and disrupted epithelial cells, which is evidenced by the pathological findings of diffuse alveolar damage with fibromyxoid exudates and macrophage infiltration in the lung tissue from patients with COVID-19.9
The limitation of this study is that induced sputum was used in the control group to compare airway mucus aspirated via bronchoscopy from patients with COVID-19, because it was very difficult for the patients with COVID-19 to expectorate sputum even with hypertonic saline solution inhalation.
The findings may suggest that increased level of MUCs in the airway mucus may contribute to the high viscosity of airway mucus and sputum retention in the small airway of patients with COVID-19, airway mucus clearance may be indicated to relieve respiratory distress, and MUC5AC may serve as a target for mucolytic agents in treating COVID-19. And MUC1-CT may serve as an indicator reflecting the severity of airway and alveolar epithelial cell damage.
This study was supported by grants from the National Key R&D Project (2016YFC0903700 and 2016YFC1304102), the National Natural Science Foundation of China (81520108001 and 81770043), and grant specific for COVID-19 study from Guangzhou Institute of Respiratory Health. The authors would like to thank Dr Kwang Chul Kim (University of Arizona) for the invaluable assistance with the manuscript.
The authors declare that there are no conflict of interests.
Source : https://onlinelibrary.wiley.com/doi/10.1002/jmv.26406
Author : Agostino Di Ciaula
February 2018
The spread of radiofrequency electromagnetic fields (RF-EMF) is rising and health effects are still under investigation. RF-EMF promote oxidative stress, a condition involved in cancer onset, in several acute and chronic diseases and in vascular homeostasis. Although some evidences are still controversial, the WHO IARC classified RF-EMF as “possible carcinogenic to humans”, and more recent studies suggested reproductive, metabolic and neurologic effects of RF-EMF, which are also able to alter bacterial antibiotic resistance. In this evolving scenario, although the biological effects of 5G communication systems are very scarcely investigated, an international action plan for the development of 5G networks has started, with a forthcoming increment in devices and density of small cells, and with the future use of millimeter waves (MMW). Preliminary observations showed that MMW increase skin temperature, alter gene expression, promote cellular proliferation and synthesis of proteins linked with oxidative stress, inflammatory and metabolic processes, could generate ocular damages, affect neuro-muscular dynamics. Further studies are needed to better and independently explore the health effects of RF-EMF in general and of MMW in particular. However, available findings seem sufficient to demonstrate the existence of biomedical effects, to invoke the precautionary principle, to define exposed subjects as potentially vulnerable and to revise existing limits. An adequate knowledge of pathophysiological mechanisms linking RF-EMF exposure to health risk should also be useful in the current clinical practice, in particular in consideration of evidences pointing to extrinsic factors as heavy contributors to cancer risk and to the progressive epidemiological growth of noncommunicable diseases.
Keywords: 5G; Cancer; MMW; Noncommunicable diseases; Prevention; RF-EMF.
https://www.sciencedirect.com/science/article/abs/pii/S1438463917308143?via%3Dihub
JOURNAL OF BIOLOGICAL REGULATORS & HOMEOSTATIC AGENTS
Vol. 34, no. 4, xx-xx (2020) – Received May 13, 2020 – Accepted June 9, 2020
M. Fioranelli¹, A. Sepehri¹, M.G. Roccia¹, M. Jafferany², O. Yu. Olisova³, K.M. Lomonosov³ and T. Lotti¹,³ ¹Department of Nuclear, Sub-nuclear and Radiation Physics, G. MarconiUniversity, Rome, Italy; ²Central Michigan Saginaw, Michigan , USA; ³Department of Dermatology and Venereology, I.M.Sechenov First Moscow State Medical University, Moscow, Russia
In this research, we show that 5G millimeter waves could be absorbed by dermatologic cells acting like antennas, transferred to other cells and play the main role in producing Coronaviruses in biological cells. DNA is built from charged electrons and atoms and has an inductor-like structure. This structure could be divided into linear, toroid and round inductors. Inductors interact with external electromagnetic waves, move and produce some extra waves within the cells. The shapes of these waves are similar to shapes of hexagonal and pentagonal bases of their DNA source. These waves produce some holes in liquids within the nucleus. To fill these holes, some extra hexagonal andpentagonal bases are produced. These bases could join to each other and form virus-like structures such as Coronavirus. To produce these viruses within a cell, it is necessary that the wavelength of external waves be shorter than the size of the cell. Thus 5G millimeter waves could be good candidates for applying in constructing virus-like structures such as Coronaviruses (COVID-19) within cells.
Coronavirus disease (COVID-19) is the main problem this year involving the entire world (1). This is an infectious disease caused by a newly-discovered coronavirus. This virus is a member of related viruses that cause diseases in mammals and birds. In humans, coronaviruses cause respiratory tract infections that can be mild, such as some cases of the common cold (among other possible causes, predominantly rhinoviruses), and others that can be lethal, such as SARS, MERS, and COVID-19. Among them, COVID-19 is an enveloped virus with a positive-sense single-stranded RNA genome and anucleocapsid of helical symmetry. The genome size of coronaviruses ranges from approximately 27 to 34 kilo bases, the largest among known RNA viruses (2, 3).
To date, many scientists have tried to find a method to cure this disease (4, 5); however, without success. COVID-19 may have effects on different types of cells. For example, it has been argued that this virus may have some effects on dermatologic cells (6). On the other hand, it has been known that some waves in 5G technology have direct effects on the skin cells (7). Thus, there are some similarities between effects of COVID-19 and waves in 5G technology.
A new question arises regarding a relationship between 5G technology and COVID-19. The 5G technology is the fifth-generation mobile technology in which its frequency spectrum could be divided into millimeter waves, mid-band, and low-band. Low- band uses a similar frequency range as the predecessor, 4G. 5G millimeter wave is the fastest, with actual speeds often being 1–2 Gbit/s down. Its frequencies are above 24 GHz, reaching up to 72 GHz, which is above the extremely high frequency band’s lower boundary. Millimeter waves have shorter range than microwaves, therefore the reactive cells are those with smaller size (8-10). Consequently, biological cells also could act like a receiver for these waves. Many researchers have considered the effects of 5G technology on human health. For example, it has been shown that 5G mobile networking technology will affect not only the skin and eyes, but will have adverse systemic effects as well (11). In another study, it was argued that 5G technologies cause great harm to human health. Cancer is only one of the many problems. 5G causes 720 (factorial) different diseases in human beings, and can kill everything that lives except some forms of microorganisms (12). To consider the effects of 5G millimeter waves on biological systems, we propose a model which describes the process of exchanging waves between 5G towers and host cells.
To date, some researchers have tried to propose a model for using waves in extracting information within cells (13, 14). These waves could be transverse electromagnetic fields or longitudinal ultrasound waves. A DNA is built from charged particles and according to laws of physics, by any motion of these particles, some electromagnetic waves emerge(15). Also, the structure of a DNA is similar to the structure of an inductor (16) in a receiver and can produce some waves. Thus, a DNA could emit some waves and interact with external waves. However, most waves have a length more than the size of cells and pass them without any effect. Only limited waves with lengths smaller than millimeter could penetrate into cell membrane and interact with DNA inductors. These wavelengths could be observed in 5G technology. Thus, towers in this technology could exchange waves with DNAs within cells and produce various types of diseases such as COVID-19. In this study, we propose a mechanism for exchanged waves between towers and host cells to obtain effective wavelengths. In our method, skin cells act as dermatologic antenna, take waves in 5G technology and transfer them to host cells. Then, DNAs within host cells interact with these waves and move. By motions of a DNA, some hexagonal and pentagonal holes emerge. To fill these holes,some bases are constructed within cells. These holes join to each other and form RNAs of COVID-19.
A mechanism for exchanging waves between towers and dermatologic cells in 5G technology.
Skin cells are in close connections with nerve fibers. These fibers in the nervous system play the role of wires which carry some electrical currents; these currents produce some electromagnetic waves. These waves and currents are taken by melanocytes, keratinocytes and other dermatologic cells and transmitted to the medium. On the other hand, skin cells could take waves of towers and transfer to other cells and neurons. Thus, dermatologic cells could act as an antenna (Fig. 1).
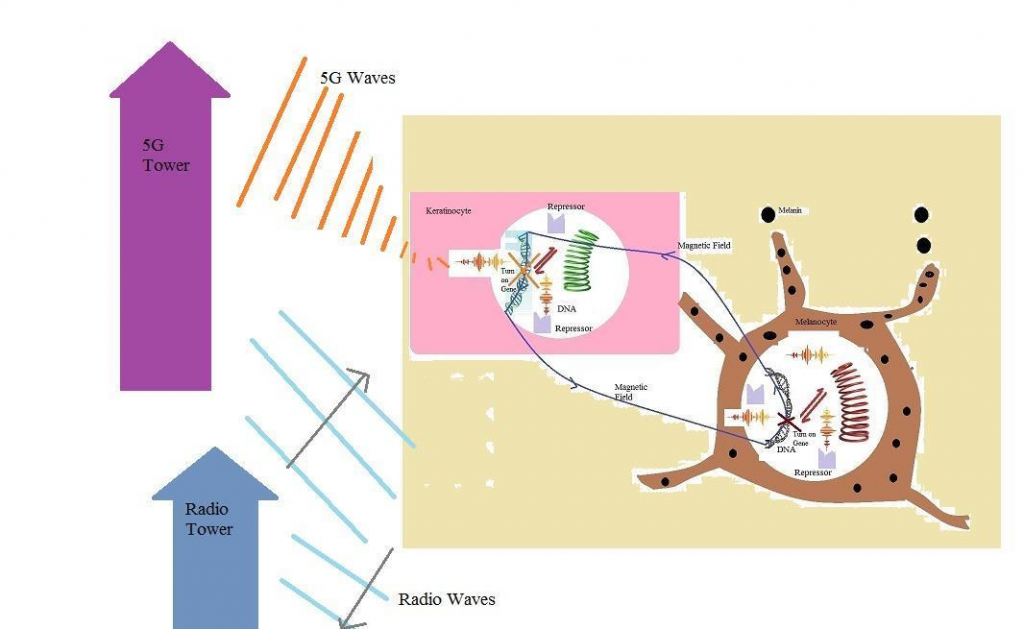
An antenna could take waves in which their wavelengths are equal to its size. Thus, millimeter waves in 5G technology could be taken more by dermatologic antennas. These waves could pass the cell membranes,enter the nucleus and interact with DNAs. Previously, it has been shown that a DNA could act as the inductor and receiver or sender of waves (16). Thus, a DNA within a dermatologic cell like a keratinocyte receives external waves and sends them to DNAs of other cells like melanocytes. Waves in 5G technology and higher technologies could contribute in gene expressions, turn on some genes and turn off others (Fig. 2).
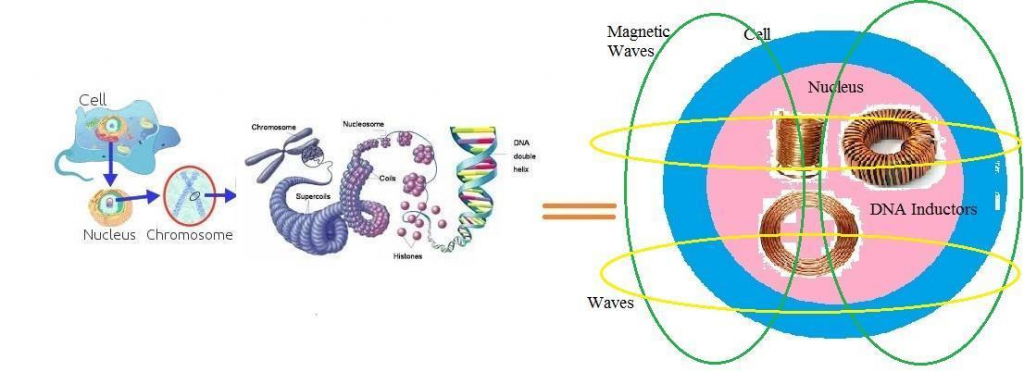
The question is whether millimeter waves in 5G technology could contribute in constructing some viruses like COVID-19 within a cell. To reply to this question, we should consider the electronic structure of a DNA and its emitted waves. A DNA is built from atoms and electrons. These particles have some electrical charges and emit electrical fields. Also, by each motion of a DNA, its atoms and electrons move. According to the laws of physics, by motion of charged particles, some magnetic waves emerge. Consequently, a DNA emits both electrical and magnetic fields and plays the role of electrical devices within a cell. The structure of a DNA within a cell is similar to the structure of an inductor. When a DNA coils around a nucleosome, it takes the shape of a toroid inductor. Also, by coiling around another axes, a DNA becomes very similar to round inductors (Fig. 3).
Fig. 1. Some waves in 5G technology could be taken by dermatologic antennas, however radio waves could not pass the skin cells
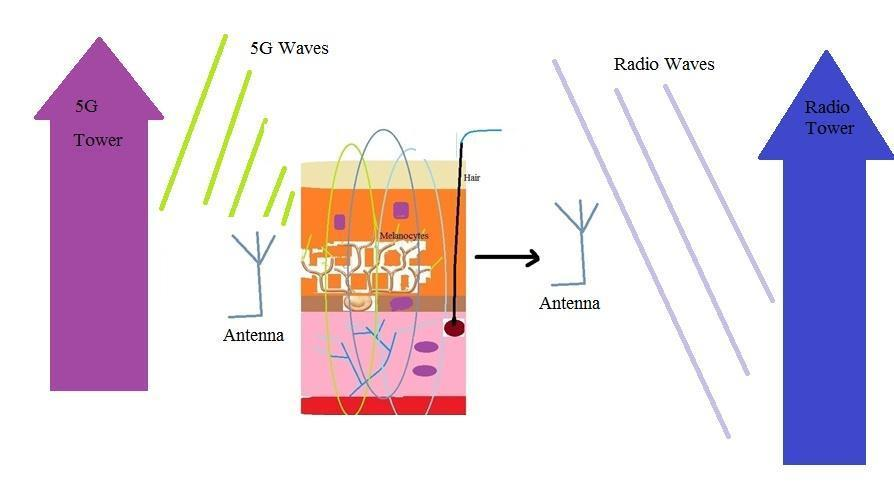
Fig. 2. Waves in 5G technology pass the cell membranes and contribute to gene expressions

Fig. 3. A similarity between different states of DNA with different types of inductors
A DNA coils several times around different axes within chromosomes and produces different types of inductors and electronic devices. Thus, any state of a DNA is similar to a type of an inductor and emits a special wave. Some of these waves are linear, some are curved and others have toroidal shapes (Fig. 4).
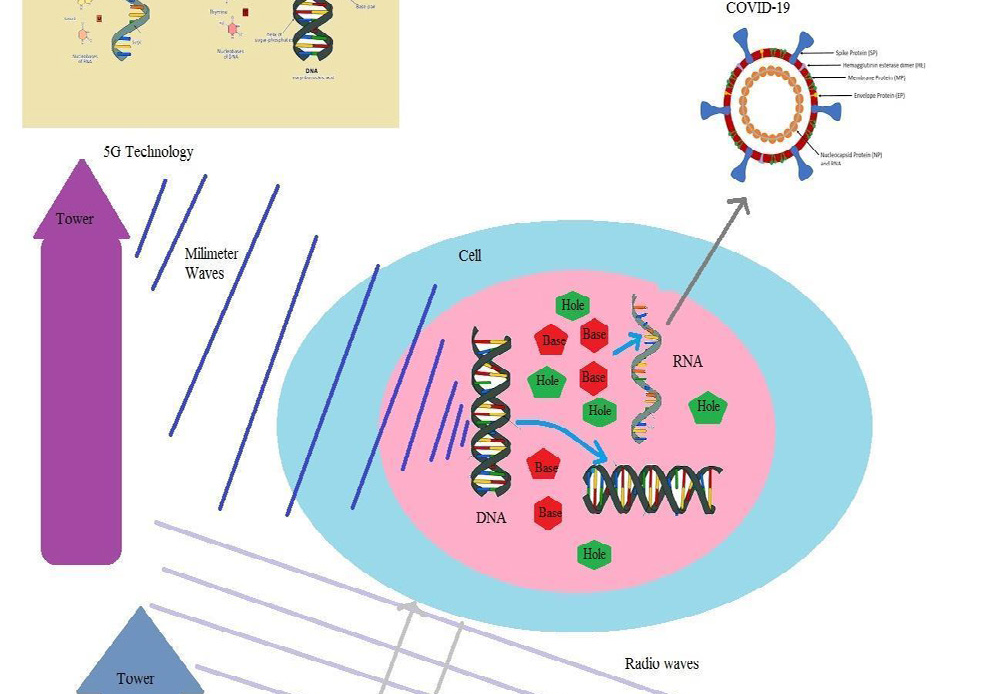
A DNA, as an electronic device within a cell, could exchange waves with medium, especially when an electromagnetic wave passes the cell membrane and the nuclear membrane, it induces an extra magnetic field within the DNA inductor and interacts with its fields. This interaction causes extra motions of this DNA, and through the motion of this DNA, its charges move and emit electromagnetic waves. The wavelength of emitted waves from a DNA is equal or less than its size within a cell. Also, shapes of radiated waves by a DNA have direct relations with the shapes of their genetic source. A DNA is formed from hexagonal and pentagonal manifolds; thus,its emitted waves have hexagonal and pentagonal shapes. These waves produce hexagonal and pentagonal holes within the liquids of a nucleus and a cell. To fill these holes, hexagonal and pentagonal molecules are built. These extra hexagonal and pentagonal bases may join to each other and form structures like RNAs of COVID-19 viruses. To produce these viruses, it is necessary that the wave lengths of external electromagnetic fields be equal or less than the size of a cell. For this reason, 5G technology waves could have the main role in the emergence of COVID-19, however radio waves could not have any effect on the evolutions within a cell (Fig. 5).
Effective wavelengths within a cell in 5G technology We propose a model to obtain a probability for the amount of effects of external fields on the evolutions of cells within a cell. This probability is related to the number of micro states of a DNA within a cell:PDNA = ΩDNA, EM / ΩDNA, tot (1)
Where ΩDNA is the probability, ΩDNA, EM is the number of micro states which are produced by the interaction between DNAs and electromagnetic waves, and ΩDNA, tot is the total number of micro states. These micro states have direct relations with entropies:
SDNA = KS LOG (ΩDNA, EM) (2)
Where SDNA is the entropy and KS is a constant. On the other hand, entropies have direct relations with energies:
SDNA =EDNA / Tcell (3)
Where EDNA is the excited energy of a DNA and Tcell is the temperature within a cell. Excited energy of a DNA depends on the linear and curved energies of hexagonal and pentagonal bases:
EDNA = UB, linear,5 V B, linear,5 + UB, curved,5 V B, curved,5 + UB, supercoil,5 V B, supercoil,5 + UB, linear,6 V B, linear,6 + UB, curved,6 V B, curved,6 + UB, supercoil,6 V B, supercoil,6 (4)
Fig. 4. A DNA within the nucleus acts as the inductor and emits magnetic waves
Where UB, linear,5/6 is the energy density of a pentagonal/hexagonal molecule, V B, linear, ,5/6 is the volume of a pentagonal/hexagonal disk , UB, curved, ,5/6 is the energy density of a pentagonal/hexagonal molecule which coils around a nucleosome, V B, curved, ,5/6 is the volume of a coiled pentagonal/hexagonal
disk, UB, supercoil, ,5/6 is the energy density of a pentagonal/hexagonal molecule which coils around supercoil axes and V B, supercoil, ,5/6 is its volume. Volumes can be obtained from the following equations:
V B, linear,5 = 5 [1/2 (rbase + xEM )2 cos (ϴpenta)sin(ϴpenta)][ rbase + xEM]
V B, linear,6 = 5 [1/2 (rbase + xEM )2 cos (ϴhexa)sin(ϴhexa)] [ rbase + xEM]
V B, curved,5 = 5π [1/2 (rbase + xEM )2 cos (ϴpenta) sin(ϴpenta)]×
[ rbase + xEM][ rhistone + xEM]2 V B, curved,6 = 5π [1/2 (rbase + xEM )2 cos (ϴhexa)sin(ϴhexa)] )]× [ rbase + xEM][ rhistone + xEM]2
V B, supercoil,5 = 5π2 [1/2 (rbase + xEM )2 cos (ϴpenta) sin(ϴpenta)]× [ rbase + xEM][ rhistone + xEM]2 [ rsupercoil + xEM]2
V B, supercoil,6 = 5π2 [1/2 (rbase + xEM )2 cos (ϴhexa)sin(ϴhexa)] )]× [ rbase + xEM][ rhistone + xEM] 2 [ rsupercoil + xEM]2 (5)
Fig. 5. 5G technology waves could pass the cell membranes and lead to production of COVID-19; however the size of radio waves are more than the size of cells and thus radio waves could not pass the cell membranes.
Where rbase is the length of a base (~10-9), rhistone is the radius of histones (~10-8), rsupercoil is the radius of a supercoil (~ 10-7), ϴhexa (π/6) is the central angle of a hexagonal molecule, ϴpenta (π/5) is the central angle of pentagonal molecule, xEM is the oscillating length which has a direct relation with the wavelength of
external field:
EEM =1/2 KEM xEM 2 = h υEM = h c/ λEM (6)
Where υEM is the frequency, λEM is the wavelength of external field, c is the velocity of light and h is the plank constant. Thus, we can write the following
equation: xEM ~ λEM -1/2 (7)
We should then calculate magnetic energies and magnetic fields. We assume that a DNA acts like an inductor and thus, we write the following equation for its magnetic fields: For linear inductor:
BDNA, linear,5/6=µ0 ngene5/6 Igene,5/6 (8)
For curved inductor:
BDNA, curved,5/6=µ0 ngene5/6 Igene,5/6/2π[rhistone + λEM-1/2] (9)
For supercoils:
BDNA, curved,5/6=µ0ngene5/6 Igene,5/6/4π2 [rsupercoil + λEM -1/2]
Where ngene5/6 is the density of genes including hexagonal and pentagonal molecules (17) within DNAs, rhistone is the size of histone (3 × 10-10) (18), rsupercoil is the radius of supercoil (~ 10-9) and Igene,5/6 is there current which moves along pentagonal/hexagonal molecules of genes. We assume that each gene is in fact a long wire that is coiled around the axis of a DNA. A DNA may have 50,000 or more gene (Ngene) (17) and each gene is around 10-12 meter long (Lgene) within a cell. Thus, we can calculate density of genes (ngene):
ngene, 5/6 =Ngene/Lgene5/6 (11)
Ngene = 50000 (17) (12)
Lgene= 10-12m (19, 20) (13)
Lgene, 5/6 = 2 ×10-12 m (19, 20) (14) ngene, 5/6 = 2.5 × 1016 (15)
To calculate the value of the current along genes, we should calculate the total effective charge of all genes (Qgene,5/6) and their velocity (Vgene,5/6).
Igene,5/6 = Qgene,5/6 Vgene,5/6 (16) Effective charges of all genes are different from their normal total charges. A gene may have a few normal charges because its charges cancel the effect of each other in the static state. However, during the gene expression and DNA evolutions, each charge has a separate effect. For this reason, we should regard total charges of all genes. To obtain this charge, we should write:
Qgene,5/6 = Ngene,5/6 qgene,5/6 (17)
Where Ngene,5/6 =2 Ngene is the number of genes including pentagonal/hexagonal molecules and qgene,5/6 is the effective charge of pentagonal/hexagonal molecules in a gene. Again, we insist that effective charge of a gene is different from its normal charge. In fact, we should regard all electrons and atoms that contribute in gene expression. For this reason, we should write:
qgene,5/6 =4Nbase qbase (18)
where Nbase is the number of base pairs within a gene (17, 18) and qbase is the effective electrical charge of a base. We can put approximate numbers and obtain the effective charge of all genes:
Nbase= 109 (21, 22) (19)
qbase =(10-20) qelectron=(10-20)× 1/6 × 10-19 (20)
Qgene, 5/6 =4 ×10-4 (21)
Now, we calculate the effective velocity of genes:
Vgene, 5/6 = Lgene, 5/6 ωgene, 5/6 (22)
This velocity depends on the length of a gene
(Lgene, 5/6) and its rotating velocity (ωgene, 5/6).
Lgene, 5/6 = 2×10-12 m (19, 20) (23)
The rotating velocity of a gene (ωgene, 5/6) can be obtained by summing over rotating velocities of all its effective charges (ωcharge, 5/6):
ωgene, 5/6 = ncharge, 5/6 ωcharge, 5/6 (24) To obtain the number of charges, we multiply number of bases and number of atoms/electrons
ncharge, 5/6 = 2Nbase Natom (25)
Now, we put approximate values for numbers and obtain velocity of genes:
Nbase= 109 (21, 22) (26)
Natom =10 (27)
ncharge, 5/6 = 2 ×1010 (28)
ωcharge , 5/6 = 2π/Tcharge, 5/6 (29)
Tcharge, 5/6 =[ λEM]1/2 /c (30)
ωcharge, 5/6 =6.28 ×10 (31)
Vgene, 5/6 =2.516 × 100 (32)
Substituting values of velocity from equation (32) and charges from equation (21) in equation (16), we can obtain the current of genes:
Igene, 5/6 ~ 10-3 (33)
Putting the current from the above equation (33) and density of genes from equation (15) in equations (6-10), we calculate magnetic fields of a DNA within a cell.
BDNA, linear,5/6~ 107
[ λEM]-1/2 (34)
BDNA, curved,5/6~ 1016 [ λEM]-1 (35)
BDNA, supercoil,5/6~ 1025 [ λEM]3//2 (36)
Using these fields, we can obtain energy density of magnetic fields around a DNA within a cell.
µ0
=4π× 10-7 (37)
UB, linear,5/6 =( [BDNA, linear,5/6] 2
/2 µ0
) ~1021 [ λEM]
-1 (38)
UB, curved,5/6 =( [BDNA, curved,5/6] 2
/2 µ0
) ~1038 [ λEM]-2
(39)
UB, supercoil ,5/6 =( [BDNA, supercoil,5/6] 2
/2 µ0 ) ~1056 [ λEM] 3 (40)
Consequently, substituting above results in equation (4), total energy can be obtained from the following equation:
EDNA = [5 [1/2 (rbase + λEM -1/2)2 cos (ϴpenta)sin(ϴpenta)] [ rbase + λEM -1/2]
5 [1/2 (rbase + λEM -1/2 )2 cos (ϴhexa)sin(ϴhexa)][ rbase
λEM -1/2]] × 1021 [ λEM]-1 +[ 5π [1/2 (rbase + λEM -1/2)2 cos (ϴpenta)sin(ϴpenta)]× [ rbase + λEM -1/2][ rhistone + λEM -1/2]2
5π [1/2 (rbase + λEM -1/2 )2 cos (ϴhexa)sin(ϴhexa)] )]× [ rbase + xEM][ rhistone + λEM -1/2]2 × 1038 [ λEM]-2
[ 5π2 [1/2 (rbase + λEM -1/2 )2 cos (ϴpenta)sin(ϴpenta)]× [ rbase + xEM][ rhistone + λEM -1/2]2 [ rsupercoil + λEM -1/2]2
5π2 [1/2 (rbase + λEM -1/2)2 cos (ϴhexa)sin(ϴhexa)] )]× [ rbase + λEM - 1/2][ rhistone + λEM -1/2]2 [ rsupercoil + λEM -1/2]2 ] × 1056 [ λEM]-3 (41)
Substituting the above equation in equations (1- 3), we can obtain the probability for the amount of effects of external fields on the evolutions of DNAs
within a cell:
PDNA = exp (KS
EDNA / Tcell) / ΩDNA, tot (42)
Fig. 6. The probability of the effect of waves on the evolutions of a DNA within a cell in terms of wavelength The above probability depends on the wavelength of external fields.

In Fig. 6, we show the probability for producing hexagonal and pentagonal DNA holes within a cell. This figure indicates that by decreasing the wavelength (< 10⁻³m), waves pass the cell membrane and interact with DNAs. This interaction causes the motions of DNAs. By motions of DNAs, their charges move and emit strong waves. These waves produce hexagonal and pentagonal holes within a cell. To fill these holes, extra bases are produced. These bases could join to each other and form viruses such as COVID-19.
Our results show that, by decreasing the wave length, waves emitted from towers in 5G and higher technologies could have more effect on evolutions of DNAs within cells. This is because dermatologic cell membranes act as an antenna for these waves. They are built from charged particles,such as electrons and atoms, and could emit or receive waves. On the other hand, an antenna could only take waves in which their lengths are not greater than its size. Thus, a cell membrane could take millimeter waves in 5G technology. These waves could pass the membrane and interact with biological matters within a cell.
If wavelengths of 5G waves be equal or less than the size of a nucleus, they can pass the nuclear membrane and interact with DNAs. These DNAs are built from hexagonal and pentagonal bases and, by their motions, some holes emerge. These holes are filled by hexagonal and pentagonal extra bases which are constructed by cells. These bases could join to each other and form some viruses such as Coronavirus. It is concluded that in the next generation of mobile technology, emitted waves of towers will have more effects on biological cells.
In this research, we have shown that new generation mobile technology, like 5G, could have the main role in constructing various types of viruses, such as Coronaviruses, within a cell. Some wavelengths in these technologies are smaller than the size of biological cells and could pass the cell membrane and enter the nucleus. These waves could be taken by dermatologic antenna, transfer to host cells, interact with DNAs and move them. A DNA is formed from charged particles and, by its motions, electromagnetic waves emerge. These waves produce hexagonal and pentagonal holes in liquids within nucleus and the cell. To fill these holes, bases are produced. These bases join to each other and can construct viruses like Corona viruses.
Originally Published : https://www.biolifesas.org/biolife/category/journals/journal-of-biological-regulators-and-homeostatic-agents/
Originally Accepted : https://pubmed.ncbi.nlm.nih.gov/32668870/
The direct targets of extremely low and microwave frequency range electromagnetic fields (EMFs) in producing non-thermal effects have not been clearly established. However, studies in the literature, reviewed here, provide substantial support for such direct targets.
Twenty-three studies have shown that voltage-gated calcium channels (VGCCs) produce these and other EMF effects, such that the L-type or other VGCC blockers block or greatly lower diverse EMF effects. Furthermore, the voltage-gated properties of these channels may provide biophysically plausible mechanisms for EMF biological effects. Downstream responses of such EMF exposures may be mediated through Ca2+/calmodulin stimulation of nitric oxide synthesis. Potentially, physiological/therapeutic responses may be largely as a result of nitric oxide-cGMP-protein kinase G pathway stimulation.
A well-studied example of such an apparent therapeutic response, EMF stimulation of bone growth, appears to work along this pathway. However, pathophysiological responses to EMFs may be as a result of nitric oxide-peroxynitrite-oxidative stress pathway of action. A single such well-documented example, EMF induction of DNA single-strand breaks in cells, as measured by alkaline comet assays, is reviewed here. Such single-strand breaks are known to be produced through the action of this pathway.
Data on the mechanism of EMF induction of such breaks are limited; what data are available support this proposed mechanism. Other Ca2+-mediated regulatory changes, independent of nitric oxide, may also have roles.
This article reviews, then, a substantially supported set of targets, VGCCs, whose stimulation produces non-thermal EMF responses by humans/higher animals with downstream effects involving Ca2+/calmodulin-dependent nitric oxide increases, which may explain therapeutic and pathophysiological effects.
How do EMFs composed of low-energy photons produce non-thermal biological changes, both pathophysiological and, in some cases, potentially therapeutic, in humans and higher animals? It may be surprising that the answer to this question has been hiding in plain sight in the scientific literature. However, in this era of highly focused and highly specialized science, few of us have the time to read the relevant literature, let alone organize the information found within it in useful and critical ways.
This study shows that:
It may be asked why we have evidence for involvement of VGCCs in response to EMF exposure, but no similar evidence for involvement of voltage-gated sodium channels? Perhaps, the reason is that there are many important biological effects produced in increased intracellular Ca2+, including but not limited to nitric oxide elevation, but much fewer are produced by elevated Na+.
The possible role of peroxynitrite as opposed to protein kinase G in producing pathophysiological responses to EMF exposure raises the question of whether there are practical approaches to avoiding such responses? Typically peroxynitrite levels can be highly elevated when both of its precursors, nitric oxide and superoxide, are high. Consequently, agents that lower nitric oxide synthase activity and agents that raise superoxide dismutases (SODs, the enzymes that degrade superoxide) such as phenolics and other Nrf2 activators that induce SOD activity [101], as well as calcium channel blockers may be useful. Having said that, this is a complex area, where other approaches should be considered, as well.
Although the various EMF exposures as well as static electrical field exposures can act to change the electrical voltage-gradient across the plasma membrane and may, therefore, be expected to stimulate VGCCs through their voltage-gated properties, it may be surprising that static magnetic fields also act to activate VGCCs because static magnetic fields do not induce electrical changes on static objects. However, cells are far from static. Such phenomena as cell ruffling [102],[103] may be relevant, where thin cytoplasmic sheets bounded on both sides by plasma membrane move rapidly. Such rapid movement of the electrically conducting cytoplasm, may be expected to influence the electrical charge across the plasma membrane, thus potentially stimulating the VGCCs.
Earlier modelling of electrical effects across plasma membranes of EMF exposures suggested that such electrical effects were likely to be too small to explain EMF effects at levels reported to produce biological changes (see, for example [22]). However, more recent and presumably more biologically plausible modelling have suggested that such electrical effects may be much more substantial [104–109] and may, therefore, act to directly stimulate VGCCs.
Direct stimulation of VGCCs by partial depolarization across the plasma membrane is suggested by the following observations discussed in this review:
One question that is not answered by any of the available data is whether what is known as ‘dirty electricity’ [111–113], generated by rapid, in many cases, square wave transients in EMF exposure, also acts by stimulating VGCCs. Such dirty electricity is inherent in any digital technology because digital technology is based on the use of such square wave transients and it may, therefore, be of special concern in this digital era, but there have been no tests of such dirty electricity that determine whether VGCCs have roles in response to such fields, to my knowledge. The nanosecond pulses, which are essentially very brief, but high-intensity dirty electricity do act, at least in part, via VGCC stimulation (Table 1), suggesting that dirty electricity may do likewise. Clearly, we need direct study of this question.
The only detailed alternative to the mechanism of non-thermal EMF effects discussed here, to my knowledge, is the hypothesis of Friedman et al. [114] and supported by Desai et al. [115] where the apparent initial response to EMF exposure was proposed to be NADH oxidase activation, leading to oxidative stress and downstream regulatory effects. Although they provide some correlative evidence for a possible role of NADH oxidase [114], the only causal evidence is based on a presumed specific inhibitor of NADH oxidase, diphenyleneiodonium (DPI). However, DPI has been shown to be a non-specific cation channel blocker [116], clearly showing a lack of such specificity and suggesting that it may act, in part, as a VGCC blocker. Consequently, a causal role for NADH oxidase in responses to EMF exposure must be considered to be undocumented.
In summary, the non-thermal actions of EMFs composed of low-energy photons have been a great puzzle, because such photons are insufficiently energetic to directly influence the chemistry of cells. The current review provides support for a pathway of the biological action of ultralow frequency and microwave EMFs, nanosecond pulses and static electrical or magnetic fields: EMF activation of VGCCs leads to rapid elevation of intracellular Ca2+, nitric oxide and in some cases at least, peroxynitrite. Potentially therapeutic effects may be mediated through the Ca2+/nitric oxide/cGMP/protein kinase G pathway. Pathophysiological effects may be mediated through the Ca2+/nitric oxide/peroxynitrite pathway. Other Ca2+-mediated effects may have roles as well, as suggested by Xu et al. [26].
Full Paper = https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3780531/